You take your medication for pain, expecting relief. Instead, the pain gets worse. It spreads beyond the original site, becoming more sensitive to touch or temperature changes that previously didn't bother you. This isn't just tolerance-it might be Opioid-Induced Hyperalgesia, a condition where opioids paradoxically increase sensitivity to pain. Known as OIH, this phenomenon affects approximately 2-15% of chronic opioid users and represents a critical challenge in modern pain management.
First documented in 1971 by Eddy et al., who observed that repeated morphine injections produced heightened pain responses in rats, OIH has evolved from an experimental curiosity to a recognized clinical reality. The Palliative Care Network of Wisconsin's Fast Fact #142, published in September 2024, describes OIH as a state of nociceptive sensitization caused by opioid exposure, distinct from disease progression or pharmacological tolerance. Understanding this condition is crucial because it explains why some patients experience worsening pain despite escalating doses-a scenario that can lead to dangerous dose increases rather than appropriate treatment adjustments.
Understanding the Mechanisms Behind Opioid-Induced Hyperalgesia
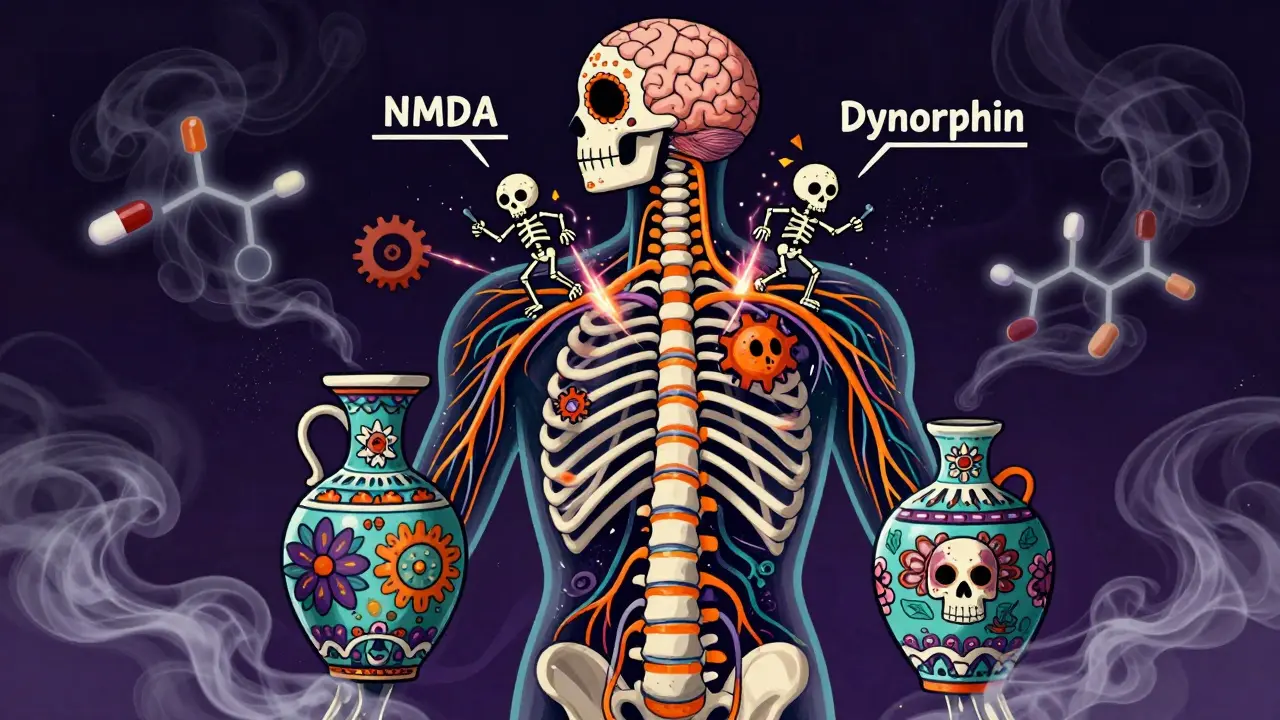
The precise molecular mechanisms driving OIH remain incompletely understood, but research points to several interconnected neurobiological pathways. The most widely accepted theory involves central sensitization through activation of N-methyl-D-aspartate (NMDA) receptors in the central nervous system. When opioids bind to their receptors, they inadvertently trigger these NMDA receptors, leading to enhanced pain signaling rather than suppression.
Additional contributing factors include toxic effects of opioid metabolites-particularly morphine-3-glucuronide and hydromorphone-3-glucuronide-which accumulate especially in patients with renal impairment. Increased spinal dynorphin activity plays another role, as does enhanced descending facilitation from the rostral ventromedial medulla. Genetic influences related to catechol-O-methyltransferase (COMT) enzyme activity also appear significant, explaining why some individuals develop OIH while others don't, even under similar dosing conditions.
Dr. J. David Clark, Professor of Anesthesiology at Stanford University, emphasizes that "OIH represents a fundamental neuroadaptive change in pain processing that requires clinicians to rethink traditional approaches to opioid dose escalation." This perspective highlights how our understanding of pain modulation continues evolving, moving beyond simple receptor blockade models toward complex neural network interactions.
| Mechanism | Description | Clinical Relevance |
|---|---|---|
| NMDA Receptor Activation | Central sensitization through opioid-triggered NMDA receptor activation | Explains efficacy of NMDA antagonists like ketamine |
| Opioid Metabolite Toxicity | Accumulation of morphine-3-glucuronide and similar compounds | Particularly relevant in renal failure patients |
| Spinal Dynorphins | Increased activity of endogenous pain-enhancing peptides | Contributes to diffuse pain patterns |
| Genetic Factors (COMT) | Variations in catechol-O-methyltransferase enzyme activity | Explains individual susceptibility differences |
Recognizing Opioid-Induced Hyperalgesia: Clinical Presentation
Clinically, OIH manifests through specific characteristics that distinguish it from other causes of worsening pain. Patients typically report increasing sensitivity to painful stimuli (hyperalgesia), worsening pain despite escalating opioid doses, and pain that becomes more diffuse, extending beyond the original distribution pattern. According to the Palliative Care Network of Wisconsin (2024), patients may develop allodynia-pain from normally non-painful stimuli-that appears unassociated with their original pain condition.
OIH can occur at any opioid dose but emerges more frequently with high parenteral doses of morphine (typically exceeding 300mg/day) or hydromorphone, particularly in patients with compromised kidney function where metabolites accumulate. The USC Orofacial Pain and Oral Medicine Wiki (2023) specifies that OIH intensity correlates with both dose escalation and duration of exposure, with symptoms typically appearing after 2-8 weeks of continuous therapy in susceptible individuals.
Distinguishing OIH from opioid tolerance presents a significant diagnostic challenge. While tolerance requires dose escalation to maintain analgesic effect, OIH features pain worsening with dose increases. Both conditions often coexist, creating a confusing clinical picture. Dr. Stephan Schug, a leading pain specialist at the University of Sydney, cautions that "OIH should not be the first diagnosis in any case, even if the patient has been on long-term opioid treatment," advocating instead for careful differential diagnosis.
Quantitative sensory testing (QST) offers valuable diagnostic assistance by measuring pain thresholds before and after opioid administration. Patients experiencing OIH typically demonstrate decreased pain thresholds (increased sensitivity) following opioid exposure, providing objective evidence supporting clinical suspicion. However, QST remains primarily a research tool rather than routine clinical practice due to accessibility limitations.
Differential Diagnosis: Distinguishing OIH from Other Causes
Recognition of OIH requires meticulous clinical assessment to exclude alternative explanations for worsening pain. Clinicians should suspect OIH when opioid treatment effectiveness diminishes without corresponding disease progression, particularly when accompanied by unexplained pain reports, diffuse allodynia unrelated to the original condition, and increased pain levels correlating with escalating doses.
The Palliative Care Network of Wisconsin (2024) emphasizes that OIH constitutes a diagnosis of exclusion, necessitating thorough evaluation to rule out other potential causes. Common mimics include disease progression, inadequate dosing, development of new pain generators, psychological factors amplifying pain perception, and withdrawal symptoms during dose reductions.
Diagnostic challenges stem from OIH's variable presentation across patients and the difficulty distinguishing it from tolerance. Some experts estimate that OIH may account for 10-30% of cases previously attributed solely to tolerance, highlighting the importance of considering this possibility in refractory pain scenarios. The Opioid-Induced Hyperalgesia Questionnaire (OIHQ), validated in a 2017 study published in the Journal of Pain Research, demonstrates 85% sensitivity and 78% specificity, offering a structured approach to identifying potential OIH cases.
- Disease Progression: Evaluate underlying pathology through imaging, laboratory tests, and clinical examination to confirm stability or advancement.
- Tolerance Development: Assess whether previous dose escalations provided temporary relief before requiring further increases.
- Withdrawal Symptoms: Consider timing relative to dose changes and presence of characteristic withdrawal signs such as sweating, nausea, or anxiety.
- New Pain Sources: Conduct comprehensive physical examination to identify additional anatomical contributors to pain complaints.
- Psychological Amplification: Screen for depression, anxiety, catastrophizing, or other cognitive-behavioral factors influencing pain perception.

Management Strategies for Opioid-Induced Hyperalgesia
Effective management of OIH focuses on modifying opioid therapy rather than continuing dose escalation. Primary treatment approaches involve reducing the opioid dosage, tapering opioids completely, or switching to alternative agents with different metabolic profiles. The Palliative Care Network of Wisconsin (2024) specifically recommends decreasing the opioid dose by 10-25% every 2-3 days or transitioning to a different opioid class.
Methadone frequently proves effective in treating OIH due to its unique pharmacological properties, including NMDA receptor antagonist activity alongside traditional mu-opioid receptor agonism. This dual mechanism addresses both the primary pain pathway and the sensitized NMDA receptors contributing to hyperalgesia. Sub-anesthetic doses of ketamine (typically 0.1-0.5 mg/kg/hour) have demonstrated efficacy in reversing OIH by directly blocking NMDA receptors, though practical implementation requires careful monitoring and specialized expertise.
Other pharmacological options target central sensitization pathways through alpha-2 agonists like clonidine (0.1-0.3 mg twice daily) and gabapentinoids such as gabapentin (300-1800 mg three times daily). These medications provide complementary mechanisms addressing neuropathic components of OIH while allowing gradual opioid reduction. Non-pharmacological interventions, including cognitive behavioral therapy and physical therapy, form essential components of multimodal pain management strategies.
Implementation challenges commonly arise from patient resistance to opioid dose reduction, reported in 40-60% of cases according to clinical observations by the Palliative Care Network of Wisconsin. Effective communication about OIH mechanisms and expected outcomes helps overcome this barrier. Successful management typically requires 2-4 weeks of dose adjustment to observe initial clinical improvement, with complete resolution often taking 4-8 weeks.
Current Developments and Future Directions
The landscape of OIH recognition and management continues evolving rapidly. Recent developments include the 2024 publication of Fast Fact #142 by the Palliative Care Network of Wisconsin, providing updated clinical guidance reflecting current evidence. Ongoing NIH-funded research (NCT05217891, expected completion 2026) investigates genetic markers for OIH susceptibility, potentially enabling personalized prevention strategies.
Future directions encompass development of genetic testing panels to identify patients harboring COMT polymorphisms predisposing them to OIH. Two commercial tests are anticipated for launch in Q2 2025, according to Genetic Engineering & Biotechnology News (2024). The American Pain Society's 2024 research priorities designate OIH as a "high-priority target" for novel analgesic development, with pharmaceutical investment in OIH-specific treatments increasing by 27% year-over-year.
Market context reveals growing awareness within pain management communities. The global opioid analgesics market reached $21.8 billion in 2023 (Statista, 2024), with approximately 191 million opioid prescriptions dispensed in the United States in 2022 (CDC, 2023). Industry trends show increasing emphasis on OIH education, with 78% of pain management fellowships now incorporating OIH into their curriculum (American Board of Pain Medicine, 2023).
Regulatory considerations include the FDA's 2022 requirement for updated labeling on long-acting opioids to include information regarding OIH as a potential adverse effect. Competitive dynamics feature pharmaceutical companies developing NMDA receptor modulators, with three compounds currently undergoing Phase II/III trials specifically targeting OIH (ClinicalTrials.gov, 2024). Adoption rates among pain specialists have risen from approximately 30% in 2010 to 65% in 2024, based on surveys conducted by the American Academy of Pain Medicine (AAPM, 2024).
What is the difference between opioid tolerance and opioid-induced hyperalgesia?
Opioid tolerance requires increasing doses to maintain the same level of pain relief, whereas opioid-induced hyperalgesia (OIH) causes pain to worsen despite dose increases. Tolerance reflects diminished response to the drug itself, while OIH represents heightened sensitivity to pain stimuli triggered by opioid exposure. Both conditions can coexist, making differentiation clinically challenging.
How common is opioid-induced hyperalgesia among chronic pain patients?
Clinical studies indicate that OIH affects approximately 2-15% of chronic opioid users. Some experts suggest it may account for 10-30% of cases previously attributed solely to tolerance. Recognition rates have improved significantly, with adoption among pain specialists rising from 30% in 2010 to 65% in 2024.
Can opioid-induced hyperalgesia be reversed?
Yes, OIH is generally reversible through appropriate intervention. Treatment involves reducing opioid dosage by 10-25% every 2-3 days, switching to alternative opioids like methadone, or adding NMDA receptor modulators such as ketamine. Initial improvements typically emerge within 2-4 weeks, with complete resolution often occurring within 4-8 weeks.
Which patients are most at risk for developing OIH?
Patients receiving high parenteral doses of morphine (>300mg/day) or hydromorphone face elevated risk. Individuals with renal impairment are particularly vulnerable due to accumulation of toxic opioid metabolites. Genetic variations affecting COMT enzyme activity also influence susceptibility, though predictive testing remains largely investigational as of 2024.
What role do NMDA receptors play in opioid-induced hyperalgesia?
NMDA receptors serve as central mediators in OIH pathophysiology. Opioid exposure triggers inadvertent activation of these receptors in the central nervous system, leading to enhanced pain signaling rather than suppression. This mechanism explains why NMDA receptor antagonists like ketamine effectively treat OIH by blocking the sensitization pathway.